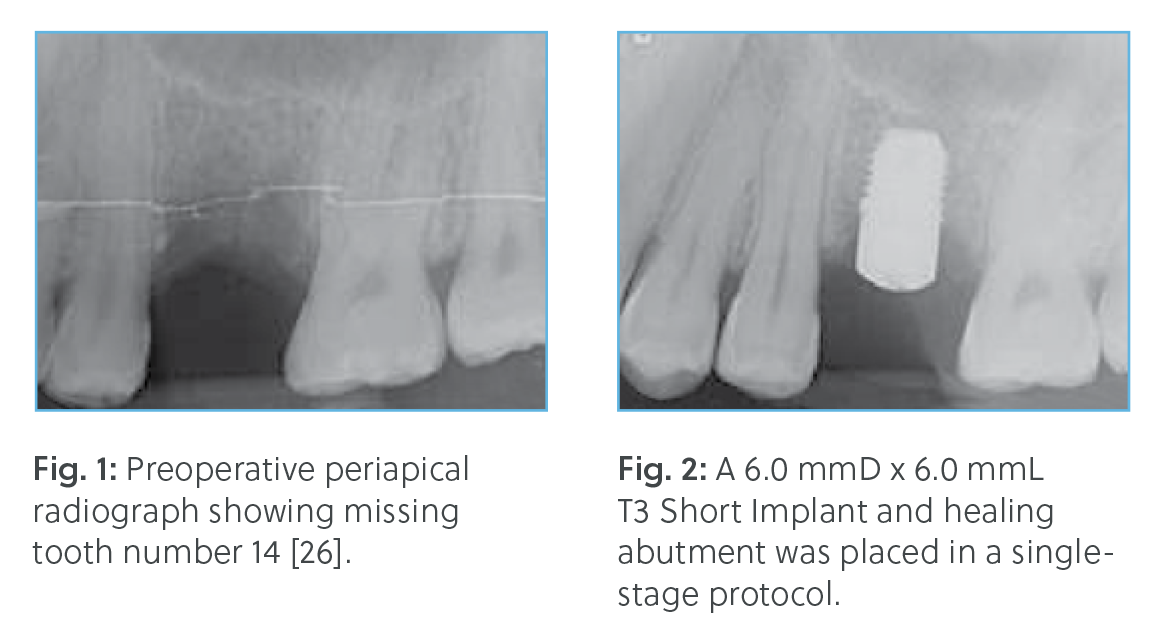
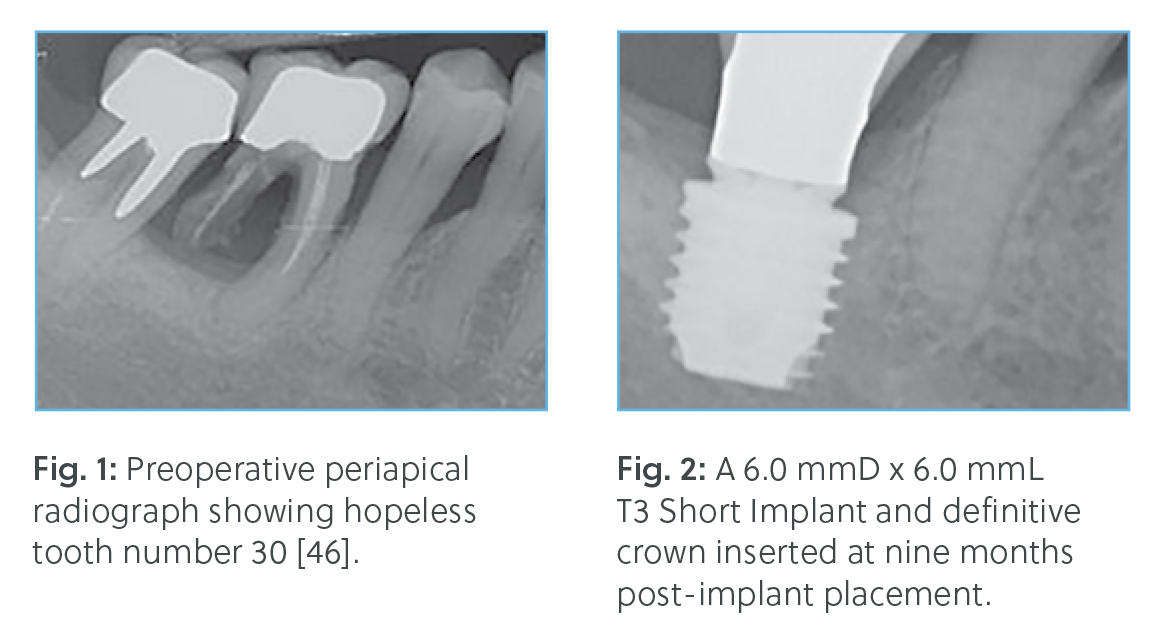
† Dr. Block, Dr. Goené, Dr. Grunder, Dr. Lazzara, Dr. Makigusa, Dr. Meltzer, Dr. Méndez, Dr.
Meyenberg, Dr. Nevins, Dr. Östman, Dr. Rodríguez, Dr. Segalá, Dr. Scutellá, Dr. Tarnow, Dr. Testori and Dr. Vela have financial relationships with Biomet 3i, LLC resulting from speaking engagements, consulting engagements and other retained services.
†† Dr. Gubbi, Mr. Suttin and Mr. Towse contributed to the above research while employed by Biomet 3i.
References 6–10 discuss the Tapered Implant macrodesign, which is incorporated into the T3 Implant. References 10–13 discuss the Osseotite and/or NanoTite Implants’ dual acid-etched or DCD technology, which is incorporated into the T3 Implant. References 20–22 discuss PREVAIL® Implants with an integrated platform switching design, which is also incorporated into the T3 Implant.