
Puros® Cortico-Cancellous Particulate
The Power Of Puros Allografts
Clinicians around the globe have counted on the Puros family of allografts for hard- and soft-tissue augmentation procedures for years.

Puros Cortico-Cancellous Particulate Allograft
An exact ratio of 70% cortical and 30% cancellous bone particulate. This mixture combines the clinical advantages of both Puros Cortical and Puros Cancellous Particulate Allograft materials.
Puros Cortical can be used in space maintenance and volume enhancement procedures.1, 2 It is slow-resorbing and maintains an open network for the proliferation of bone-forming cells.42, 44
Puros Cortico-Cancellous Particulate Clinical Evidence

- Without sacrificing ridge contour, cortical particles remodel into a dense, lamellar structure as well as viable bone – with similar density to native bone45
- 2 mm in buccal bone thickness when used in a “sandwich” technique for the treatment of localized buccal dehiscence defects45
- 40% mineralized bone and 0.47% residual grafting materials after 4 months healing time in sinus lift procedures46
- Clinical and radiographic graft stability after 5 years follow up in sinus lift procedures47
- Reduced vertical and horizontal bone resorption when used in immediate implant placement extraction sites48
Puros Allograft Block Unique Tutoplast Process
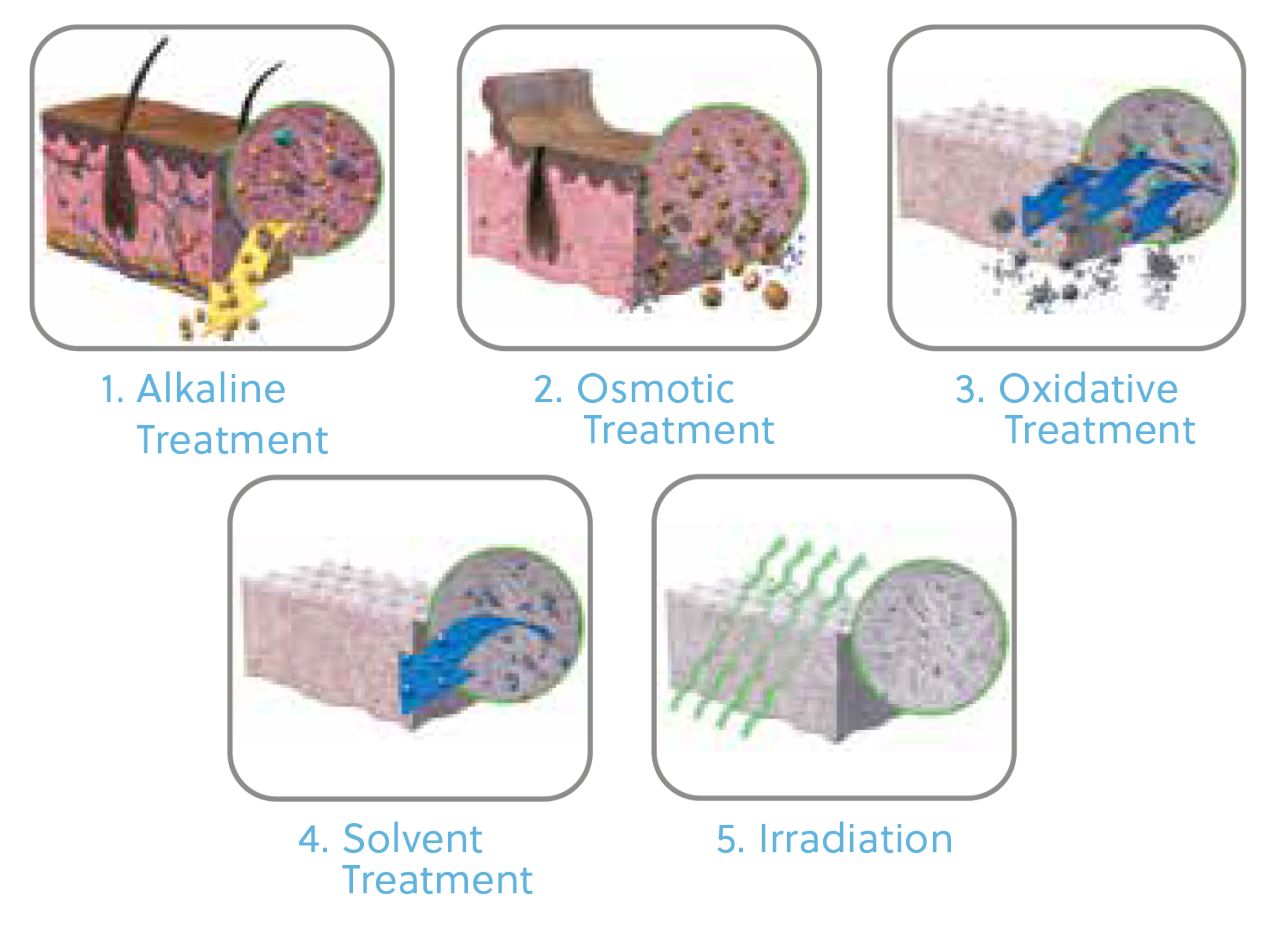
For over 50 years, a variety of Tutoplast processed tissues have been safely used in more than 11 million procedures.
Osmotic, oxidative, and alkaline (if indicated) treatments break down cell walls, inactivate pathogens, and remove bacteria. Solvent dehydration allows for room-temperature storage of tissue without damaging the native tissue structure.
Low-dose gamma irradiation ensures a sterility assurance level (SAL) of 10-6 of the final packaged graft.7
Clinically successful in procedures for:
- Sinus augmentation 44, 46,49, 50
- Alveolar ridge augmentation 43, 51, 52
- “Tenting” and “sandwich” grafting techniques 12-16
- Immediate implant post extraction sockets 48
